The 2012 Meeting of the British Crystallographic Association was held at Warwick University.┬Ā Oxford was very well represented this year with Kirsten Christensen chairing the Programme Committee and prizes being awarded to Joe Paddison, Nick Funnell and Emma McKinley.
Other contributions from Oxford include:
Karim J. Sutton, Kirsten E. Christensen, Amber L. Thompson & Richard I. Cooper
Exploiting the Tunable Wavelength Capabilities of I19 (Poster & Oral Presentation)
Callum A. Young & Andrew L. Goodwin
An Examination of the Low Temperature Structure of Fe3O4 Using Reverse Monte Carlo Refinements (Oral Presentation)
Andrew L. Goodwin
Frameworks, Flexibility and Frustration (Oral Presentation)
Kirsten E. Christensen, Amber L. Thompson & Arwen R Pearson
Discussion Forum:┬Ā From Small Molecules to Proteins:┬Ā Bridging the Gap
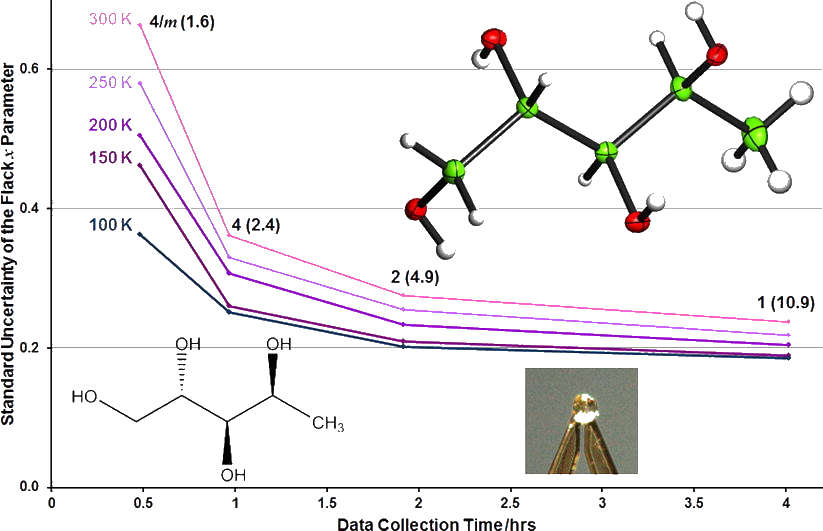
Emma J. McKinley, Kirsten E. Christensen & Amber L. Thompson
Investigation of the Transient Modulated Phase of Barluenga’s Reagent (Poster)
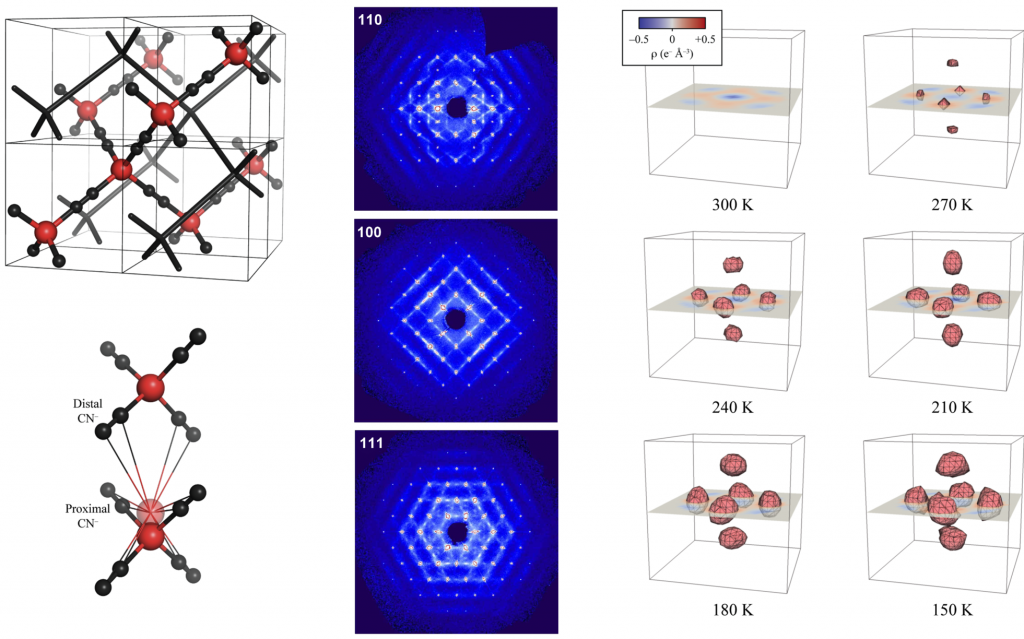
Andrew B. Cairns, Amber L. Thompson, Andrew L, Goodwin, Matthew G. Tucker & Julien Haines
Anomalous Compression of Cyanide Frameworks:┬Ā Mechanisms for Extreme Negative Response (Poster)
Callum A. Young, Edward Dixon, Michael A. Hayward & Andrew L. Goodwin
Apical Oxygen Bonding in YBCO – A Reverse Monte Carlo Approach (Poster)
Ines E. Collings, Andrew B. Cairns, Amber L. Thompson, Andrew L, Goodwin, Philip Pattison, Julien Haines, Matthew G. Tucker, Julia E. Parker & Chiu C. Tang
Transferable Mechanics in Molecular Framework Materials (Poster)
Joseph A. M. Paddison, Andrew L. Goodwin, Ross Stewart, Pascal Manuel, Matthias J. Gutmann, David A. Keen, Matthew G. Tucker, Andrew Wildes & Brian Rainford
Frustration and Magnetic Diffuse Scattering in┬Ā╬▓-Mn0.8Co0.2 and MnO:┬Ā A Reverse Monte Carlo Study (Poster)
Matthew Cliffe & Andrew L. Goodwin
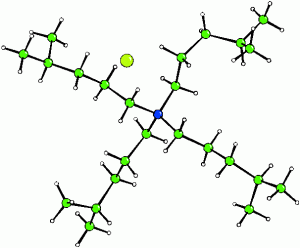
Disorder and Defects in Zirconium MOFs (Poster)
Nicholas P. Funnell, Martin T. Dove Andrew L. Goodwin Simon Parsons & Matthew G. Tucker
How Molecules Prepare to Undergo a Transition:┬Ā The Onset of Disorder in Solid Cyclohexane (Poster).

Karim speaks to the Young Crystallographers

Emma, Richard and Ines go to the conference dinner

Ines and Emma

This time Karim brought his Dad…

Callum and Emma at Dinner

Kirsten presents Nick with his prize
Emma receives her prize

<no comment />

Contemplating Fourier Transforms…